 
What is the electronic configuration of Sodium 11? What is the boiling Point of Sodium in Kelvin?īoiling Point of Sodium in Kelvin is 1156 K. 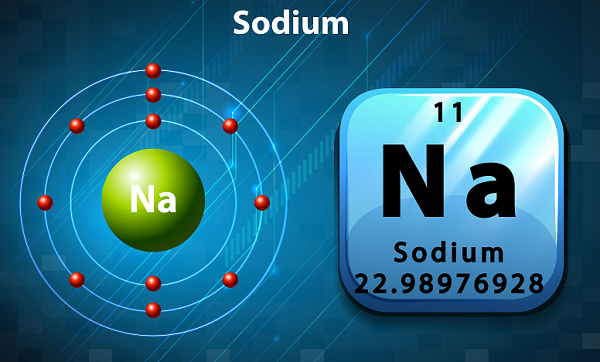
Melting Point of Sodium in Kelvin is 370.87 K. What is the melting Point of Sodium in Kelvin? What is the boiling Point of Sodium?īoiling Point of Sodium is 1156 K. Sodium has 11 electrons out of which 1 valence electrons are present in the 3s1 outer orbitals of atom. How many valence electrons does a Sodium atom have? It is located in group 1 and period 3 in the modern periodic table. 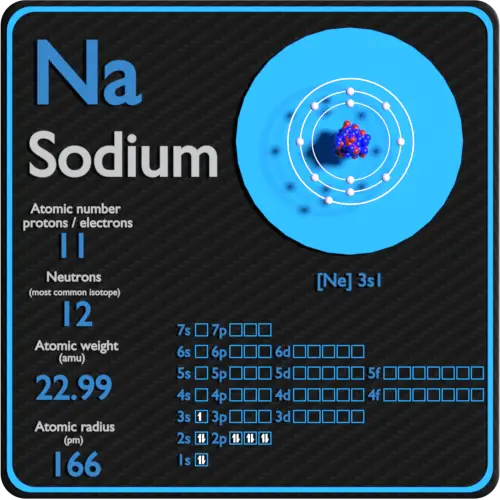
Sodium is the 11 element on the periodic table. Sodium is a chemical element with the symbol Na and atomic number 11. What is the position of Sodium in the Periodic Table? Sodium is a chemical element with symbol Na and atomic number 11. To form abbreviated notation of electronic configuration, the completely filled subshells are replaced by the noble gas of the preceding period in square brackets. The abbreviated electronic configuration of Sodium is 3s1. What is the abbreviated electronic configuration of Sodium? The electronic configuration of Sodium is 1s2 2s2 2p6 3s1. What is the electronic configuration of Sodium? Sodium Thermal Properties - Enthalpies and thermodynamics Optical Properties of Sodium Refractive IndexĪcoustic Properties of Sodium Speed of Sound Sodium Heat and Conduction Properties Thermal Conductivity Refer to table below for the Electrical properties ofSodium Electrical Conductivity Hardness of Sodium - Tests to Measure of Hardness of Element Mohs Hardness Refer to below table for Sodium Physical Properties DensityĠ.968 g/cm3(when liquid at m.p density is $0.927 g/cm3) 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
